News Releases


Amarillo, Texas, Aug. 13, 2020 (GLOBE NEWSWIRE) -- via NEWMEDIAWIRE -- Amarillo Biosciences, Inc. (“ABI” or the “Company”), (AMAR), a diversified healthcare company, today announced that it is taking steps to reaffirm its position as the world leader in low-dose non- injectable interferon research. ABI will strengthen its commitment to bring its products to market by supporting global advancement of its therapeutics with other like-minded biopharma industry partners.
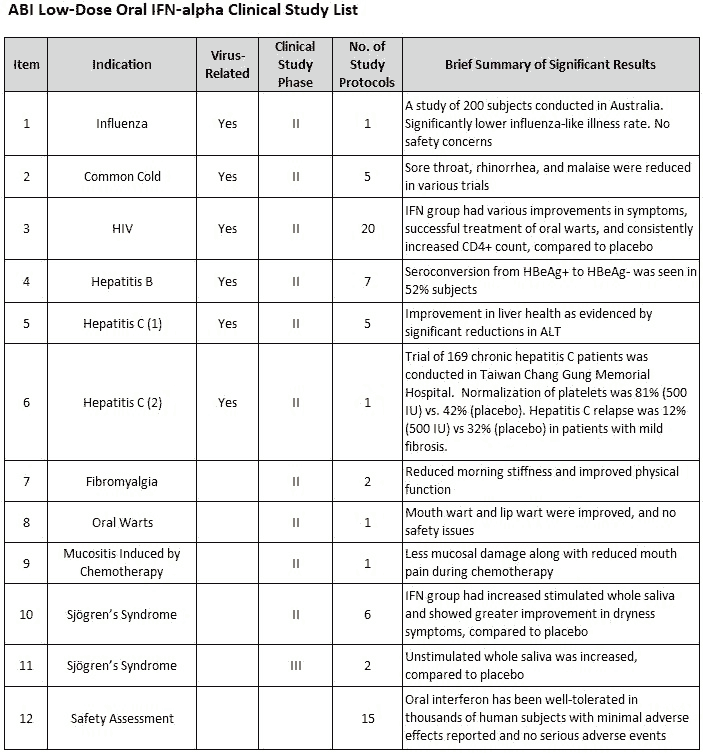
Since its inception in 1984, ABI has been the leading pioneer in the research of low-dose non- injectable interferon (IFN) delivered through nasal, oral mucosal, topical and transdermal pathways, either in liquid, ointment or lozenge form. The company’s drug development program, VELDONA (Very Low-Dose Oral Interferon Alpha), encompasses the most comprehensive library of scientific clinical data for low-dose interferon having completed more than 100 pre-clinical animal and human studies on its safety and efficacy over the course of thirty-five years. ABI also has an accomplished track record of completing well over 60 clinical trials for numerous disease indications, with many reaching phase II and phase III
trials. Historically, the company has focused on R&D involving low-dose, orally administered patented lozenges containing the natural immune system activator interferon alpha (IFN-α) as a treatment for numerous human disease indications such as influenza, hepatitis C, thrombocytopenia, HIV, SARS and other conditions ranging from genital warts to canker sores.
Since the occurrence of Covid-19, there have been no drugs approved for administration early in the stages of infection or for preventive administration to those at high risk. However, positive data from laboratory research and clinical trials using recombinant IFN, either alone or in combination with other antivirals, indicate IFN as an attractive therapeutic option. Researchers have valid concerns regarding the side effects and pro-inflammatory effects resulting from high- dose injectable IFN used in many of these recent studies. This is a debilitating problem common among high-dose IFN injectable treatments that highlights the significance of exploring low-dose IFN in further research and also underscores the importance of leveraging ABI’s extensive clinical research to expedite clinical trials.
ABI has been approached by a diverse roster of institutions seeking to collaborate in Covid-19 related research. These parties include Chinese health authorities, foreign governments, international CRO firms and researchers familiar with our work in low-dose IFN. ABI is currently in discussions with certain third-party sponsors interested in supporting the company’s late stage, clinical trial drug development program. “We are confident in securing a long-term strategic arrangement that will not only define the continuation for the origination of specially- formulated IFN product but also construct a value chain infrastructure offering drug development turnkey services. We envision the creation of a powerhouse organization to reestablish ABI as the foremost authority in the field of low-dose interferon,” said Stephen T. Chen, Ph.D., Chairman & CEO of ABI.
The company is extremely encouraged by recent positive medical research that continues to demonstrate low-dose IFN’s potential promise for protecting susceptible healthy people during the coronavirus pandemic. One compelling study, though yet to be peer reviewed, is an experimental trial of recombinant human IFN alpha nasal drops to prevent Covid-19 in medical hospital staff in Hubei Province, China. Data from this research showed that among nearly 3,000 front line nurses and doctors, a 28-day incidence of Covid-19 was zero in both the high and low- risk groups and the incidence of new-onset clinical symptoms for pneumonia was also zero. There were no adverse reactions observed.
ABI has recently assembled an experienced research management team and secured interferon sources to initiate clinical studies in the U.S., China, Taiwan and Australia. The company will seek to work with global partners to develop its VELDONA technology for the treatment and prevention of Covid-19. We have reconnected with research and medical experts that are avid supporters of the VELDONA drug program and together plan to orchestrate a concerted effort to support the advancement of various low-dose IFN formulations and delivery modalities that can help stem further spread of the pandemic. Moreover, in order to better support other independent drug developers in facilitating a quicker path to market for their own respective interferon research, ABI has decided to offer its drug technology research as an open collaboration platform for the prevention and treatment of Covid-19, along with other future viral outbreaks. The company will also out-license VELDONA with new partners to continue development of high- probability interferon therapeutics for several disease indications it has already progressed. For instance, ABI plans to resume its late-stage clinical trials for thrombocytopenia, an immune condition causing low platelets, in which the company owns several related patents to alleviate recurrence of hepatitis C in patients. Our open source platform will offer partners the ability to optimize the development process by accessing ABI’s thirty-plus years of valuable clinical data to selectively target suitable disease indications. ABI extends an open invitation to any interested organizations seeking to collaborate in this endeavor.
About Amarillo Biosciences, Inc.
Amarillo Biosciences, Inc. (ABI) is a diversified healthcare company engaged in the discovery and development of pharmaceutical and biotech products. Our goal is to introduce novel products that actively stimulate and rejuvenate the human body to combat disease and enhance the ability to heal. We are an industry leader in the advancement of low-dose non-injectable interferon as a therapeutic treatment for numerous indications such as thrombocytopenia, Sjögren's syndrome, hepatitis C virus (HCV) and influenza, a potential multi-billion dollar market opportunity. ABI primarily operates through three divisions: Pharmaceutical, Medical and Consumer. The Pharmaceutical division leverages a proprietary library of over a hundred scientific and clinical data studies on various human and animal applications of low-dose interferon, for patent licensing and commercialization opportunities with global partners. The Medical division is focused on developing an innovative, state-of-the-art technology to treat metabolism related diseases such as Type 1 and Type 2 diabetes in Asia. The Consumer division includes a range of nutraceutical and food supplement products that utilize our unique liposomal delivery systems. ABI currently has offices in the United States and Taiwan.
FORWARD-LOOKING STATEMENTS:
CERTAIN STATEMENTS MADE THROUGHOUT THIS DOCUMENT ARE "FORWARD- LOOKING STATEMENTS" WITHIN THE MEANING OF THE PRIVATE SECURITIES LITIGATION REFORM ACT OF 1995 (THE "ACT"). FORWARD-LOOKING STATEMENTS INCLUDE, WITHOUT LIMITATION, ANY STATEMENT THAT MAY PREDICT, FORECAST, INDICATE OR IMPLY FUTURE RESULTS, PERFORMANCE, ACHIEVEMENTS, COSTS OR EXPENSES AND MAY CONTAIN WORDS SUCH AS "BELIEVE," "ANTICIPATE," "EXPECT," "ESTIMATE," "PROJECT," "BUDGET," “POTENTIAL,” OR WORDS OR PHRASES OF SIMILAR MEANING. FORWARD- LOOKING STATEMENTS INVOLVE RISKS AND UNCERTAINTIES THAT MAY CAUSE ACTUAL RESULTS TO DIFFER MATERIALLY FROM THOSE PROJECTED IN THE FORWARD-LOOKING STATEMENTS.
EXCEPT FOR THE HISTORICAL INFORMATION CONTAINED HEREIN, THE MATTERS DISCUSSED IN THIS PRESS RELEASE ARE FORWARD-LOOKING STATEMENTS THAT INVOLVE RISKS AND UNCERTAINTIES. THE FOLLOWING IMPORTANT FACTORS COULD CAUSE ACTUAL RESULTS TO DIFFER MATERIALLY FROM THOSE IN THE FORWARD-LOOKING STATEMENT, INCLUDING, BUT NOT LIMITED TO, THE ANTICIPATED AVAILABILITY AND POTENTIAL BENEFITS ASSOCIATED WITH THE USE OF THE PROPOSED TREATMENTS, PRODUCT QUALITY, MANUFACTURING OR SUPPLY, OR PATIENT SAFETY ISSUES, UNCERTAINTIES RELATED TO PRODUCT DEVELOPMENT, REGULATORY AND OTHER GOVERNMENT APPROVALS UNDER INTERNATIONAL AND U.S. LAWS, THE EFFICACY OF PROPOSED TREATMENTS AND THE OUTCOME OF CLINICAL TRIALS, DEPENDENCE ON THIRD-PARTY PROPRIETARY TECHNOLOGY, INGREDIENTS, AND MATERIALS, MARKET DEMAND AND ACCEPTANCE OF ORAL INTERFERON OR THE COMPANY’S OTHER PRODUCT CANDIDATES, COMPETITIVE PRODUCTS AND TREATMENTS THAT CURRENTLY EXIST OR THAT ARE IN DEVELOPMENT, AND OTHER RISKS DETAILED FROM TIME TO TIME IN THE COMPANY’S FILINGS WITH THE SECURITIES AND EXCHANGE COMMISSION. ABI DOES NOT UNDERTAKE TO UPDATE ITS FORWARD-LOOKING STATEMENTS.
For more information contact: llin@amarbio.com, (806) 376-1741
